Products
Integrity, passion, professionalism, respect and cooperation
Address: No. 1000, North of Shengzhou Avenue, Shengzhou City, Zhejiang Province, China
Tel: +86-575-83123228
Fax: +86-575-83101736
E-mail: hr@alkpharm.cn
URL: http://www.alkpharm.com
10mg-28tablets Nifedipine Sustained-release Tablets

Nifedipine Sustained-release Tablets Insturctions
Please read the instructions carefully and use them under the guidance of physician.
[Drug name]
Common name: Nifedipine Sustained-release Tablets
English name: Nifedipine Sustained-release Tablets
Chinese Pinyin: Xiaobendiping Huanshi Pian
[Ingredients]
The active ingredient of this product is: nifedipine. Chemical name: 2,6-Dimethyl-4-(2-nitrophenyl)-1,4-dihydro-3,5-pyridinedicarboxylic acid dimethyl ester.
Structural formula: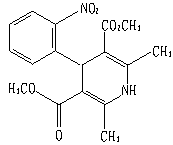
Molecular formula: C17H18N2O6
Molecular weight: 346.34
[Properties]
This product is a film-coated tablet, and it is yellow after removing the film coating.
[Indications]
Various types of hypertension and angina.
[Specifications]
10mg
[Usage and dosage]
Oral: 10~20 mg (1 to 2 tablets) at a time, 2 times a day. Extreme amount, 40mg (4 tablets) at a time, 0.12g (12 tablets) a day.
[Taboo]
Cardiogenic shock, acute myocardial infarction (within 80 days).
[Use in pregnant and lactating women]
The product is contraindicated in pregnancy
[Use in children]
The product is contraindicated in children.
[Use in the elderly]
This experiment was not performed and there is no reliable reference.
[Storage]
Store in a well-closed container, protected from light.
[Packing]
Aluminum-plastic blister packaging, 28 tablets/box, 30 tablets/box, 48 tablets/box, 60 tablets/box, plus an aluminum-plastic composite bag.
[Valid period]
24 months.
[Executive standards]
WS1-(X-056)-2004Z
[Approval Number]
State Medical Permitment No. H33020046
[Manufacturer]
Enterprise: Zhejiang Anglikang Pharmaceutical Co., Ltd.
Add: No. 1000, North of Shengzhou Avenue, Shengzhou City, Shaoxing City, Zhejiang, China
P. C.: 312400
Tel: +86-575-83108588
Fax: +86-575-83101736

 Anglikang is a modern pharmaceutical company specializing in the health of Chinese people, featuring oral cephalosporins, cardiovascular and kidney diseases.
Anglikang is a modern pharmaceutical company specializing in the health of Chinese people, featuring oral cephalosporins, cardiovascular and kidney diseases. In the future development, Anglikang will adhere to the two-wheel drive of technological transformation and product innovation, and build the company into an innovative international enterprise with strong comprehensive competitiveness in the industry.
In the future development, Anglikang will adhere to the two-wheel drive of technological transformation and product innovation, and build the company into an innovative international enterprise with strong comprehensive competitiveness in the industry. Anglikang core values: integrity, passion, professionalism, respect, cooperation
Anglikang core values: integrity, passion, professionalism, respect, cooperation Anglikang always insists on technological innovation to drive enterprise development and continuously invests a large amount of research and development funds.
Anglikang always insists on technological innovation to drive enterprise development and continuously invests a large amount of research and development funds. 

