Products
Integrity, passion, professionalism, respect and cooperation
Address: No. 1000, North of Shengzhou Avenue, Shengzhou City, Zhejiang Province, China
Tel: +86-575-83123228
Fax: +86-575-83101736
E-mail: hr@alkpharm.cn
URL: http://www.alkpharm.com
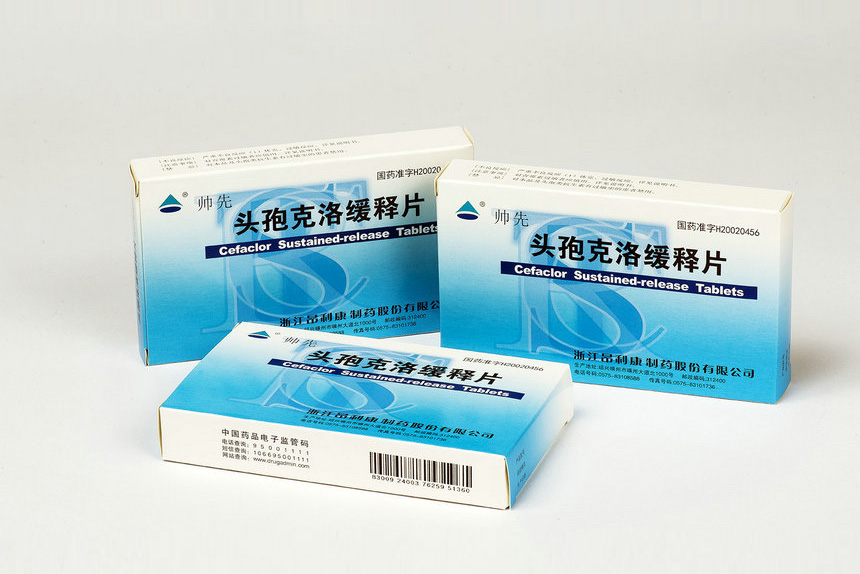
0.375g-6 tablets-1 board Cefaclor Sustained Release Tablets
Cefaclor Sustained Release Tablets Insturctions
Please read the instructions carefully and use them under the guidance of physician.
[Drug name]
Common name: Cefaclor Sustained Release Tablets
Trade name: Shuai Xian
English name: Cefaclor Sustained Release Tablets
Chinese Pinyin: ToubaoKeluo Huanshipian
[Ingredients]
The active ingredient of this product is cefaclor. Chemical name:(6R,7R)-7-[(R)-2-amino-2-phenylacetamido]-3-chloro-8-oxo-5-thia-1-azabicyclo[4.2.0] s- 2-ene-2-carboxylic acid monohydrate.
Structural formula: .png)
Molecular formula: C15H14ClN3O4S·H2O
Molecular weight: 385.82
[Properties]
This product is a film-coated tablet, which is yellowish after removal of the coating.
[Indications]
This product is indicated in light and moderate infections caused by staphylococcus aureus, hemolytic streptococcus, pneumococcus, Escherichia coli, Klebsiella, influenza bacillus, Shigella and other sensitive strains:
[Specifications]
0.375g(asC15H14ClN3O4S)
[Usage and dosage]
Oral, the usual dose for adults is 0.375g (1 tablet) each time, 2 times a day, and the dose of severe patients is doubled.
[Storage]
It is kept in closed container in cool (under 20 ° C) and dry place.
[Packing]
Aluminum-plastic blister packaging, 8 tablets×1 board / box; 6 tablets×1 board / box;6 tablets×2 boards/ box; 4 tablets×1 board / box.
[Valid period]
Tentatively twenty-four months
[Executive standards]
WS-245(X-211)-2002-2016Z
[Approval Number]
State Medical Permitment No. H20020456
[Manufacturer]
Enterprise: Zhejiang Anglikang Pharmaceutical Co., Ltd.
Production address: No. 1000, North of Shengzhou Avenue, Shengzhou City, Shaoxing City, Zhejiang, China
P. C.: 312400
Tel: +86-575-83108588
Fax: +86-575-83101736

 Anglikang is a modern pharmaceutical company specializing in the health of Chinese people, featuring oral cephalosporins, cardiovascular and kidney diseases.
Anglikang is a modern pharmaceutical company specializing in the health of Chinese people, featuring oral cephalosporins, cardiovascular and kidney diseases. In the future development, Anglikang will adhere to the two-wheel drive of technological transformation and product innovation, and build the company into an innovative international enterprise with strong comprehensive competitiveness in the industry.
In the future development, Anglikang will adhere to the two-wheel drive of technological transformation and product innovation, and build the company into an innovative international enterprise with strong comprehensive competitiveness in the industry. Anglikang core values: integrity, passion, professionalism, respect, cooperation
Anglikang core values: integrity, passion, professionalism, respect, cooperation Anglikang always insists on technological innovation to drive enterprise development and continuously invests a large amount of research and development funds.
Anglikang always insists on technological innovation to drive enterprise development and continuously invests a large amount of research and development funds. 

